Introduction
Phytochromes are a class of photoreceptors that play an important function in mediating plant responses to light. a few of the phytochrome circle of relatives, phytochrome B (phyB) is particularly essential in regulating diverse elements of plant increase and improvement. One of the key factors of phyB characteristic is its localization to particular subnuclear systems known as nuclear bodies.
Expertise in the necessities for the localization of phyB to nuclear bodies is crucial for unraveling the mechanisms underlying its regulatory features. In this article, we can delve into the characterization of the necessities for the localization of phytochrome B to nuclear our bodies, losing light at the intricate molecular tactics worried about this crucial thing of phyB.
Importance of PHYB in Plant Biology
To start with, it is crucial to provide a few context on the importance of phyB in plant biology. PhyB is a red/far-red light photoreceptor that performs a pivotal role in regulating numerous factors of plant boom and development, together with seed germination, seedling de-etiolation, color avoidance, and flowering.
The perception of light by using phyB ends in its activation and next translocation to the nucleus, in which it interacts with a large number of signaling additives to modulate gene expression and developmental responses. The subnuclear localization of phyB to nuclear our bodies is a crucial issue of its function, as it is within those structures that phyB interacts with its downstream goals and exerts its regulatory consequences.
Regulated and Dynamic Localization of phyB
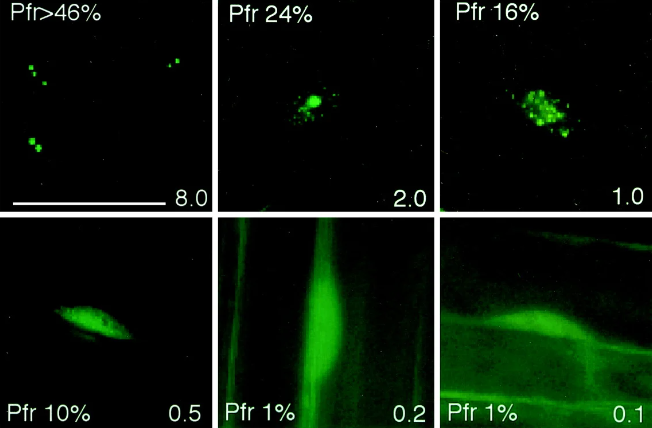
The system of phyB localization to nuclear bodies is an exceptionally regulated and dynamic phenomenon that is motivated via a spread of things. The first requirement for phyB localization to nuclear bodies is its activation by way of crimson light. Upon exposure to pink light, the inactive shape of phyB (Pr) undergoes photoconversion to the active shape (Pfr), triggering a conformational change that permits its nuclear translocation. This light-precipitated activation is a prerequisite for the following steps leading to phyB localization to nuclear bodies.
Interactions with Nuclear Proteins and Cofactors for PhyB Localization
Some other crucial requirement for phyB localization to nuclear bodies is its interaction with unique nuclear proteins and cofactors. It has been validated that phyB interacts with a selection of proteins, which include PIFs (phytochrome-interacting factors), COP1 (constitutively photomorphogenic 1), and other regulatory proteins, to modulate its nuclear localization and downstream signaling. Those interactions are essential for targeting phyB to nuclear our bodies and facilitating its practical interactions inside those subnuclear structures.
Regulation of PhyB Subnuclear Localization through Nuclear Import and Export Signals
Moreover, the subnuclear localization of phyB to nuclear bodies is influenced by the presence of unique nuclear import and export indicators inside the phyB protein. Research has recognized nuclear localization alerts (NLS) and nuclear export signals (NES) in the phyB protein that govern its dynamic shuttling between the nucleus and the cytoplasm. The appropriate law of those signals and their interactions with the nuclear import and export equipment are essential for the right localization of phyB to nuclear bodies.
Post-Translational Modifications and PhyB Localization to Nuclear Bodies
Further to protein-protein interactions and nuclear shipping alerts, the dynamic nature of phyB localization to nuclear bodies is also prompted by using post-translational changes. Phosphorylation, ubiquitination, and different adjustments were shown to modulate the subnuclear localization and stability of phyB.
For instance, the E3 ubiquitin ligase COP1 targets phyB for degradation, thereby regulating its abundance and subnuclear localization. The interplay among those post-translational modifications and their results on phyB localization to nuclear our bodies provides another layer of complexity to the regulatory mechanisms governing phyB function.
Role of Nuclear Bodies in PhyB Localization
Furthermore, the business enterprise of nuclear bodies themselves performs a critical role inside the localization of phyB. Nuclear our bodies are dynamic and heterogeneous structures that serve as websites for the meeting of various regulatory complexes and the sequestration of particular proteins.
The composition and dynamics of nuclear our bodies are motivated by means of elements along with light high-quality, developmental stage, and environmental cues, all of which can affect the subnuclear localization of phyB and its practical interactions inside these structures.
Regulatory Mechanisms of PhyB Localization to Nuclear Bodies
The characterization of the necessities for localization of phyB to nuclear our bodies has supplied valuable insights into the molecular mechanisms governing its subnuclear dynamics and regulatory capabilities.
The interplay among light signaling, protein-protein interactions, nuclear transport signals, publish-translational changes, and nuclear frame enterprise together contribute to the perfect spatiotemporal manipulation of phyB localization and activity. Information on those intricate approaches is critical for unraveling the regulatory networks that govern plant responses to light and environmental cues.
Also Read: Which Technology is Making Quantum Computing Easier to Access and Adopt?
Conclusion
In the end, the localization of phytochrome B to nuclear our bodies is a relatively regulated and dynamic manner that is essential for its regulatory functions in plant increase and improvement. The requirements for phyB localization to nuclear bodies embody a complicated interplay of light signaling, protein-protein interactions, nuclear transport signals, publish-translational modifications, and nuclear frame organization.
Characterizing those necessities has supplied treasured insights into the molecular mechanisms underlying phyB function and has paved the way for similar exploration of its regulatory roles in plant biology.
Read more about mouth larva

Harold Ayres, a captivating wordsmith and seasoned blogger, weaves tales that transport readers to worlds unknown. With his keen insight and vivid storytelling, Harold’s literary prowess captivates hearts and minds, leaving an indelible mark on the literary landscape.